Signs and symptoms at a glance
What are the early signs and symptoms of DMD?
Watch the Duchenne muscular dystrophy — Signs and symptoms animation to find out about red flag signs and characteristics of Duchenne muscular dystrophy.
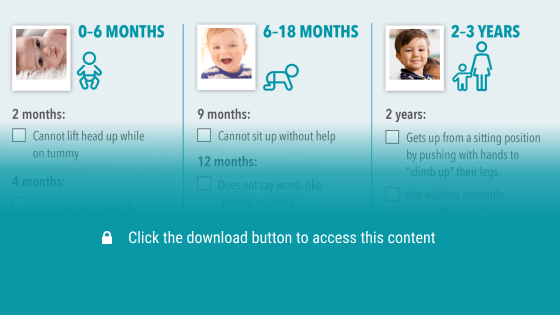
Childhood developmental milestone checklist
Recognising Duchenne muscular dystrophy signs and symptoms early is important as once muscle function is lost, it cannot be restored.1—3
Carefully monitoring baby and childhood developmental milestones can help to identify weakness earlier, allowing for early testing and diagnosis, both of which are critical to gaining access to the right treatments and services.1,4—6
The childhood developmental milestones checklist provides a summary of key motor and non-motor developmental milestones that may be missed in a child presenting with a neuromuscular disorder and children with Duchenne muscular dystrophy.
Download a copy of the developmental milestone checklistThe importance of early diagnosis
While there is no cure for DMD, studies have shown that early diagnosis and care can help slow DMD progression, and minimise the risks and complications of the disease.1,4,15—17
Prompt intervention is critical to help delay disease progression and help preserve muscle function for as long as possible, meaning that patients may:1,4,17—20
- Remain ambulatory for longer
- Preserve their pulmonary and cardiac function longer
- Have a better quality-of-life
- Live longer
Recognising DMD symptoms early also means that parents of patients can have earlier access to genetic counselling, which can assist with family planning.4
Early diagnosis may also provide the opportunity for the child to be enrolled in research-based registries and clinical trials of investigational treatments.14
Carriers of DMD: symptoms and care
Female carriers may be at risk of DMD symptoms.15,21,22
Approximately 10% of female carriers show some disease manifestations, including:15,21—24
- Muscle weakness
- Cardiomyopathy
- Central nervous system manifestations
The 2018 Duchenne Care Considerations recommend performing cardiac assessment (including an electrocardiogram and non-invasive imaging) every 3—5 years, in all female carriers in early adulthood.15 Assessments should be more frequent in those who are symptomatic or imaging-positive.15
As diagnosis of DMD in children takes an average of 2 years, from parental concern, carrier women may have more children without realising that they carry a DMD mutation.4,10,25
Learn more about Genetic counselling and Carrier screening for DMD
The symptoms of DMD in depth
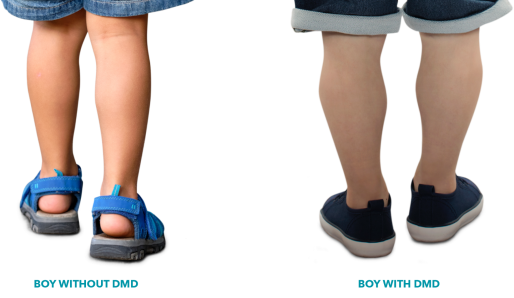
Recognising red flag signs and symptoms in DMD
Key motor and non-motor developmental milestones may be missed in a child presenting with a neuromuscular disorder and children with DMD.1 Use the checklist below to support assessment of your paediatric patients and identify missed milestones that might indicate the need to carry out a creatine kinase (CK) test.1,6
Learn more about CK testing